Sellaphora C.Mereschkowsky; 1902; 186
Key references
Mann D.G. 1989. The Diatom Genus Sellaphora: Separation from Navicula. British Phycological Journal. 24: 1-20.
Morphology
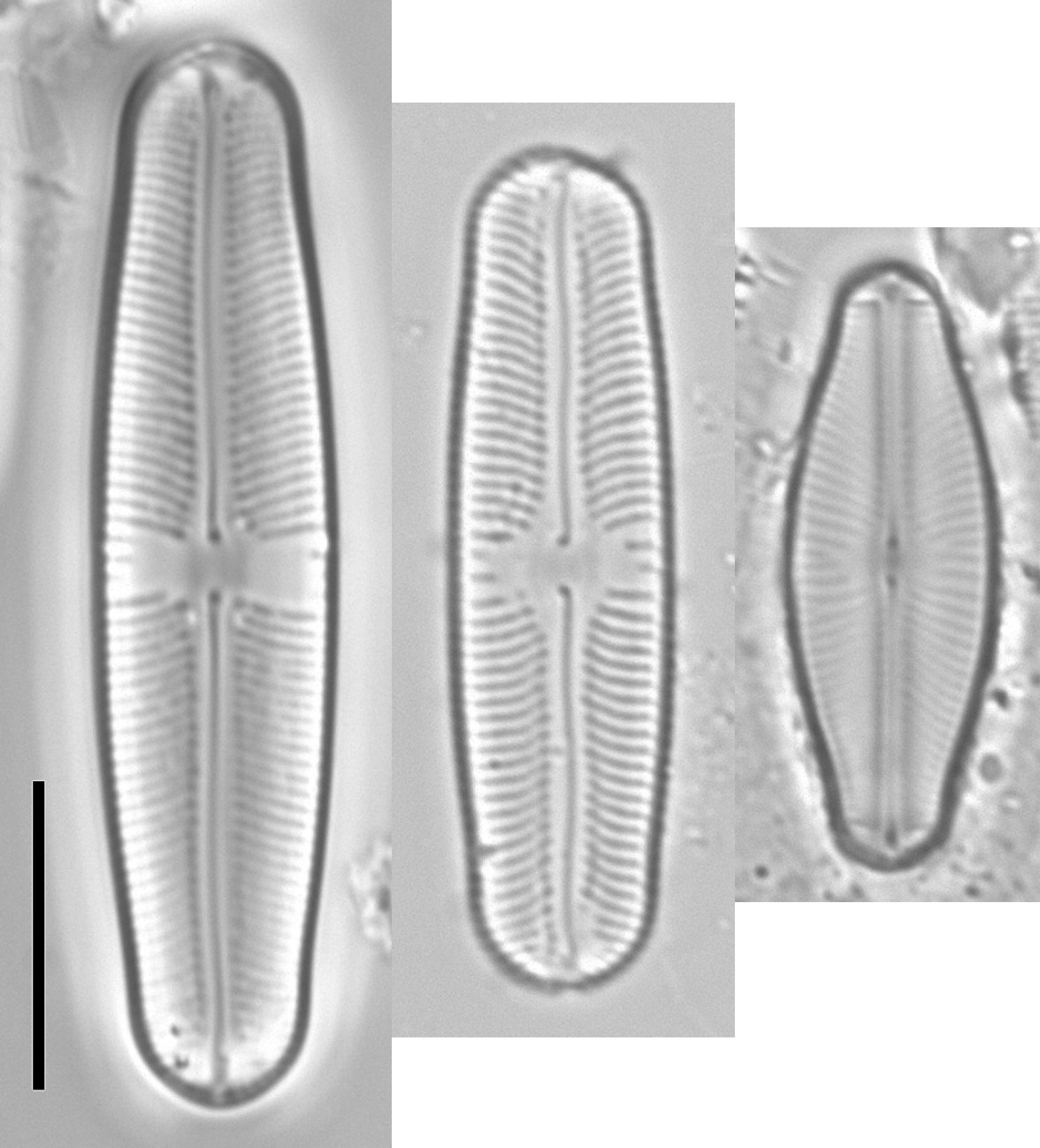
Frustules linear to lanceolate or elliptical in valve view, approximately as deep as wide and therefore quite commonly lying in girdle view; symmetrical with respect to all the principal axes and planes (i.e. isopolar, isovalvar and bilaterally symmetrical).
Valves with a flat valve face that curves gently into a moderately deep mantle. Striae usually uniseriate, rarely biseriate, running uninterrupted from the raphe-sternum to the valve margin, parallel or radiate (sometimes convergent near the valve apices, composed of small round poroids occluded by hymens).
Width of axial area variable; central area usually obvious, circular to rectangular or bow-tie-shaped, sometimes with alternately long and short striae around the central area, sometimes with ‘ghost striae’ (striae that become partially filled in during valve formation).
Raphe-sternum sometimes accompanied on either side externally by a groove, which may or may not be covered by a plain conopeum (contrast the porous conopeum of the related genus Fallacia); where present, the groove and conopeum may be interrupted in the central area or continue across it. Opposite the helictoglossa, the raphe-sternum sometimes bears transapical extensions on either side (the ‘polar bars’), which are thickened internally and appear in LM as darker lines.
Raphe central, with internal central raphe endings deflected towards the primary side of the valve. External central raphe endings usually deflected towards the primary side, usually somewhat expanded and pore-like; external polar raphe endings (terminal fissures) curved, bent or hooked towards the secondary side. There is sometimes a special larger areola at the apex of the valve, between the terminal fissure and the valve margin.
Girdle exhibiting ‘primodominance’ (Mann & Poulíčková 2019), i.e. with the first band (the valvocopula) being by far the widest of the bands, while the bands further from the valve are narrow strips or reduced to ligulae.
Many of the aspects of frustule morphology described above are symplesiomorphies, being found in a wide range of other naviculoid genera, or (in the case of the polar bars, plain conopea or the larger apical areola) are not common to all Sellaphora species. In contrast, the redescription of Sellaphora by Mann (1989) emphasized a number of non-frustular characters, viz. plastid single, H-shaped, lying with its centre against the epivalve; pyrenoid ± tetrahedral during interphase, invaginated, one per plastid, lying to one side of the cell; plastid translocated onto the girdle before mitosis; division of plastid beginning before cytokinesis (and indeed before the previous division is complete ...), proceeding asymmetrically, and arresting before completion to produce the H-shape of the chloroplast during interphase; 90º rotation of the plastid onto the epivalve occurring at the end of valve formation; gametangia not surrounded by a mucilage sheath, closely associated throughout meiosis and plasmogamy, and opening only partially to allow gamete migration via a copulation aperture; one functional gamete and one apochlorotic cell formed per gametangium; gametes physiologically anisogamous, with the auxospore being formed within one gametangium and expanding parallel to its apical axis. These characters have not been scored for all species but some appear currently to be synapomorphies, e.g. the single eccentric invaginated pyrenoid and the features of gametogenesis and fertilization. Many species possess prominent volutin granules, one in each apical vacuole.
The genus has grown enormously during the last 30 years, from 8 species when the genus was resurrected by Mann (1989) to over 250 in 2020. This growth has been partly through transfers of species from ‘Navicula’, partly through description of new species, many of them small-celled species with few obvious characters in SEM. Whether all of these species actually belong to Sellaphora will remain unclear until they are studied alive and/or with DNA sequence data.
Valves with a flat valve face that curves gently into a moderately deep mantle. Striae usually uniseriate, rarely biseriate, running uninterrupted from the raphe-sternum to the valve margin, parallel or radiate (sometimes convergent near the valve apices, composed of small round poroids occluded by hymens).
Width of axial area variable; central area usually obvious, circular to rectangular or bow-tie-shaped, sometimes with alternately long and short striae around the central area, sometimes with ‘ghost striae’ (striae that become partially filled in during valve formation).
Raphe-sternum sometimes accompanied on either side externally by a groove, which may or may not be covered by a plain conopeum (contrast the porous conopeum of the related genus Fallacia); where present, the groove and conopeum may be interrupted in the central area or continue across it. Opposite the helictoglossa, the raphe-sternum sometimes bears transapical extensions on either side (the ‘polar bars’), which are thickened internally and appear in LM as darker lines.
Raphe central, with internal central raphe endings deflected towards the primary side of the valve. External central raphe endings usually deflected towards the primary side, usually somewhat expanded and pore-like; external polar raphe endings (terminal fissures) curved, bent or hooked towards the secondary side. There is sometimes a special larger areola at the apex of the valve, between the terminal fissure and the valve margin.
Girdle exhibiting ‘primodominance’ (Mann & Poulíčková 2019), i.e. with the first band (the valvocopula) being by far the widest of the bands, while the bands further from the valve are narrow strips or reduced to ligulae.
Many of the aspects of frustule morphology described above are symplesiomorphies, being found in a wide range of other naviculoid genera, or (in the case of the polar bars, plain conopea or the larger apical areola) are not common to all Sellaphora species. In contrast, the redescription of Sellaphora by Mann (1989) emphasized a number of non-frustular characters, viz. plastid single, H-shaped, lying with its centre against the epivalve; pyrenoid ± tetrahedral during interphase, invaginated, one per plastid, lying to one side of the cell; plastid translocated onto the girdle before mitosis; division of plastid beginning before cytokinesis (and indeed before the previous division is complete ...), proceeding asymmetrically, and arresting before completion to produce the H-shape of the chloroplast during interphase; 90º rotation of the plastid onto the epivalve occurring at the end of valve formation; gametangia not surrounded by a mucilage sheath, closely associated throughout meiosis and plasmogamy, and opening only partially to allow gamete migration via a copulation aperture; one functional gamete and one apochlorotic cell formed per gametangium; gametes physiologically anisogamous, with the auxospore being formed within one gametangium and expanding parallel to its apical axis. These characters have not been scored for all species but some appear currently to be synapomorphies, e.g. the single eccentric invaginated pyrenoid and the features of gametogenesis and fertilization. Many species possess prominent volutin granules, one in each apical vacuole.
The genus has grown enormously during the last 30 years, from 8 species when the genus was resurrected by Mann (1989) to over 250 in 2020. This growth has been partly through transfers of species from ‘Navicula’, partly through description of new species, many of them small-celled species with few obvious characters in SEM. Whether all of these species actually belong to Sellaphora will remain unclear until they are studied alive and/or with DNA sequence data.
Literature
References are given in chronological order.
Reference |
Citation |
|---|---|
| Mereschkowsky C. 1902. On Sellaphora, a new genus of Diatoms. Annals and Magazine of Natural History, series 7, 9. 185-195; pl. IV. | Morphology; Taxonomy; Description; Illustrations |
| Mann D.G. 1989. The Diatom Genus Sellaphora: Separation from Navicula. British Phycological Journal. 24: 1-20. | Morphology; Taxonomy; Illustrations; |
| Lange-Bertalot H., Moser G. 1994. Brachysira. Monographie der Gattung. Bibliotheca Diatomologica 29. J. Cramer, Berlin. 212 pp | Morphology |
| Metzeltin D., Lange-Bertalot H. 2007. Tropical diatoms of South America II. Special remarks on biogeography disjunction. Iconographia Diatomologica, Vol. 18., Annotated Diatom Micrographs. Diversity-Taxonomy-Biogeography. H. Lange-Bertalot (ed.), A.R.G. Gantner Verlag K.G. 877 pp | Morphology |
| Evans K.M., Wortley A.H., Mann D.G. 2007. An assessment of potential diatom ‘barcode’ genes (cox1, rbcL, 18S and ITS rDNA) and their effectiveness in determining relationships in Sellaphora (Bacillariophyta). Protist . 158: 349–364. | Biology |
| Vanormelingen P., Evans K.M., Chepurnov V.A., Vyverman W., Mann D.G. 2013. Molecular species discovery in the diatom Sellaphora and its congruence with mating trials. Fottea, Olomouc . 13(2): 133–148. | Biology |
| Mann D.G., Poulíčková A. 2019. Homothallism, morphology and phylogenetic position of a new species of Sellaphora (Bacillariophyta), S. pausariae. Plant Ecology and Evolution . 152: 203-218. | Morphology; Biology; Illustrations; Taxonomy |
This page should be cited as:
Mann D. G. Sellaphora C.Mereschkowsky; 1902; 186. In: Jüttner I., Carter C., Cox E.J., Ector L., Jones V., Kelly M.G., Kennedy B., Mann D.G., Turner J. A., Van de Vijver B., Wetzel C.E., Williams D.M..
Freshwater Diatom Flora of Britain and Ireland. Amgueddfa Cymru - National Museum Wales. Available online at https://naturalhistory.museumwales.ac.uk/diatoms/browsespecies.php?-recid=3739. [Accessed:
].
Record last modified: 27/12/2020